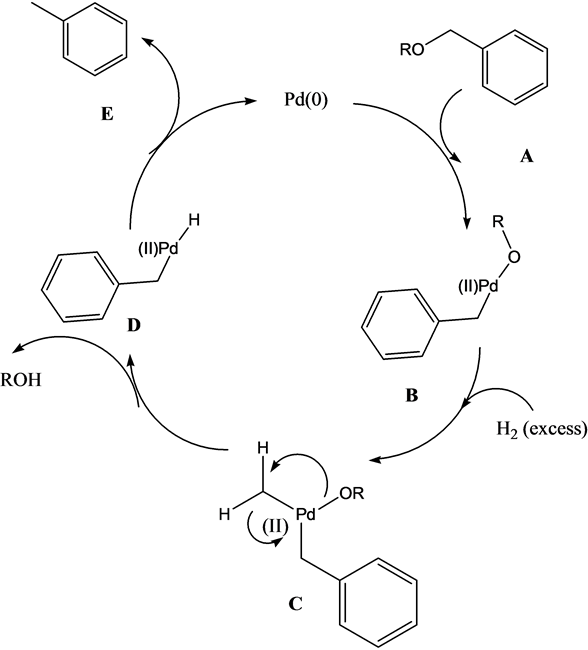SciELO - Brazil - Recent Progress in the Use of Pd-Catalyzed C-C Cross-Coupling Reactions in the Synthesis of Pharmaceutical Compounds Recent Progress in the Use of Pd-Catalyzed C-C Cross-Coupling Reactions in the

Mild Palladium‐Catalyzed Cyanation of Unprotected 2‐Iodoglycals in Aqueous Media as Versatile Tool to Access Diverse C2‐Glycoanalogues - Malinowski - 2020 - Advanced Synthesis & Catalysis - Wiley Online Library
![PDF] Palladium-triggered deprotection chemistry for protein activation in living cells. | Semantic Scholar PDF] Palladium-triggered deprotection chemistry for protein activation in living cells. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/3ae70a978b403098bfa58487a5f2c84d3e816087/2-Figure1-1.png)
PDF] Palladium-triggered deprotection chemistry for protein activation in living cells. | Semantic Scholar

Palladium-triggered deprotection chemistry for protein activation in living cells | Nature Chemistry

Irradiation-induced palladium-catalyzed decarboxylative desaturation enabled by a dual ligand system | Nature Communications
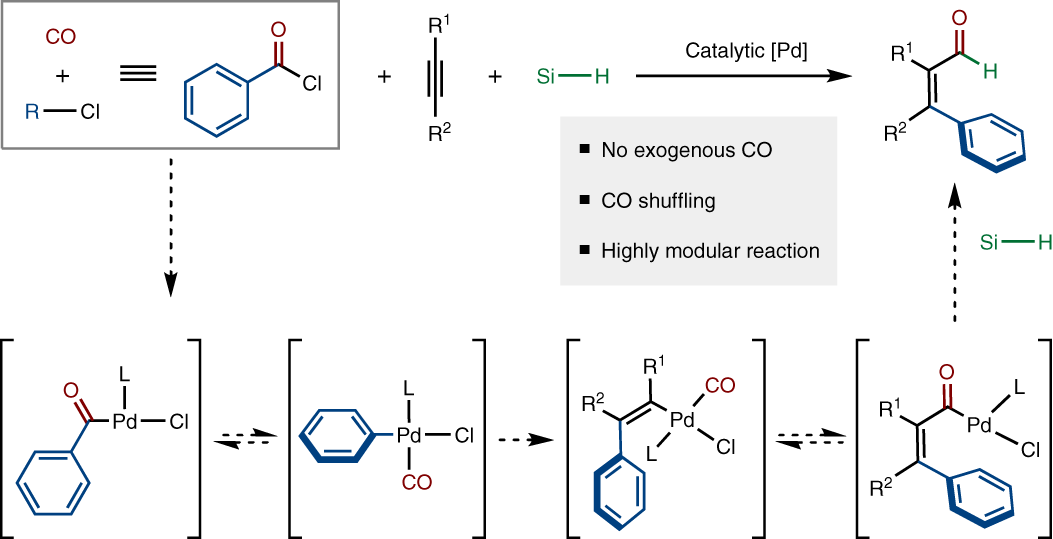
Palladium-catalysed carboformylation of alkynes using acid chlorides as a dual carbon monoxide and carbon source | Nature Chemistry

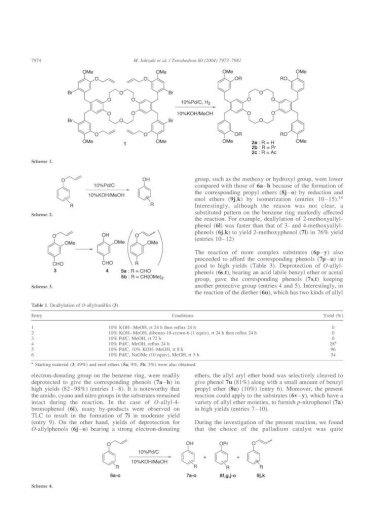
A Mild Deprotection Strategy for Allyl-Protecting Groups and Its Implications in Sequence Specific Dendrimer Synthesis
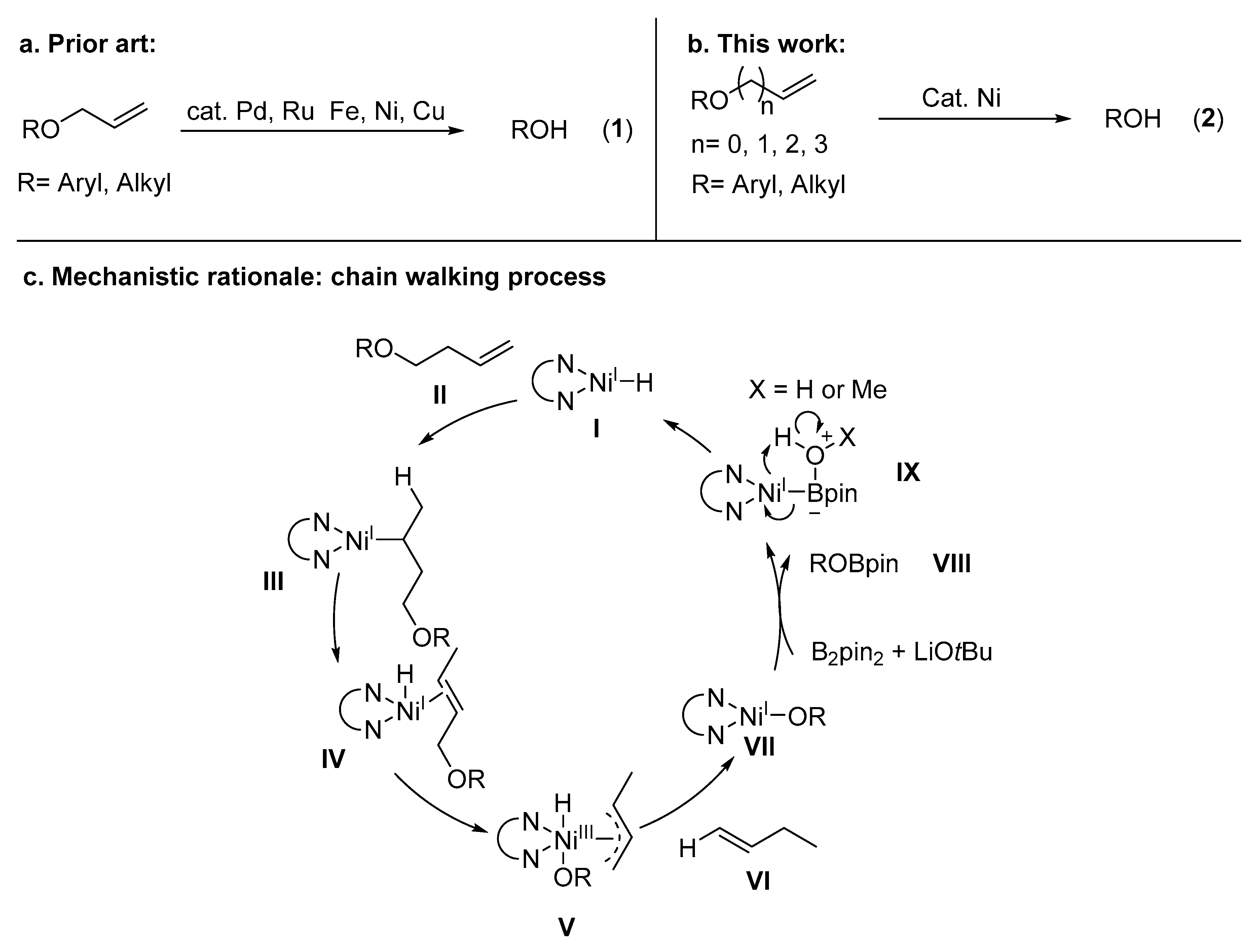
Molecules | Free Full-Text | Nickel-Catalyzed Removal of Alkene Protecting Group of Phenols, Alcohols via Chain Walking Process | HTML
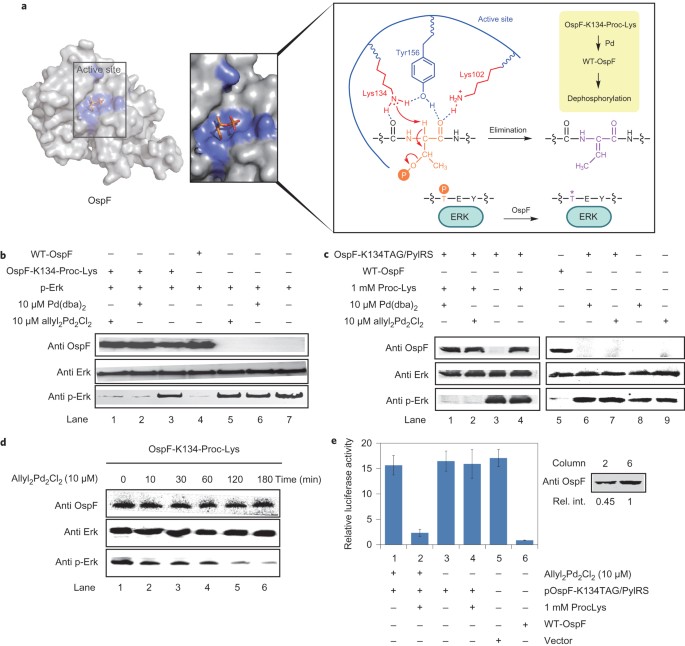
Palladium-triggered deprotection chemistry for protein activation in living cells | Nature Chemistry
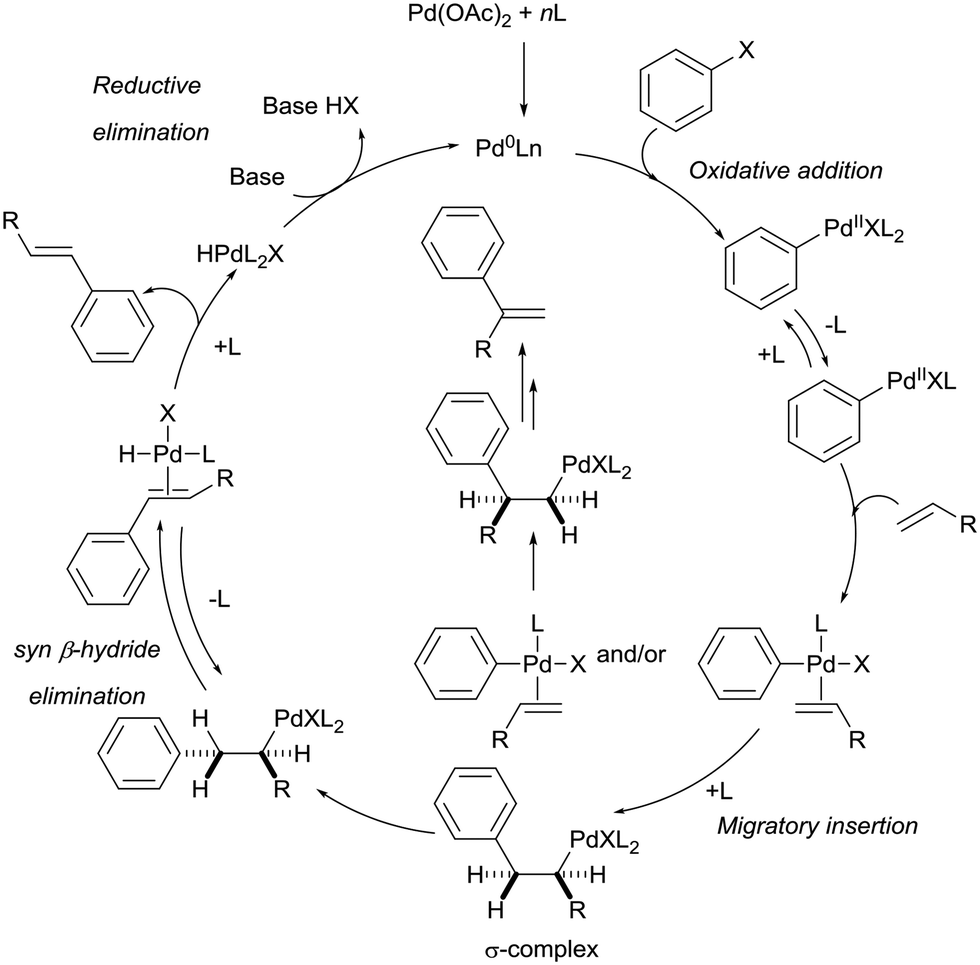
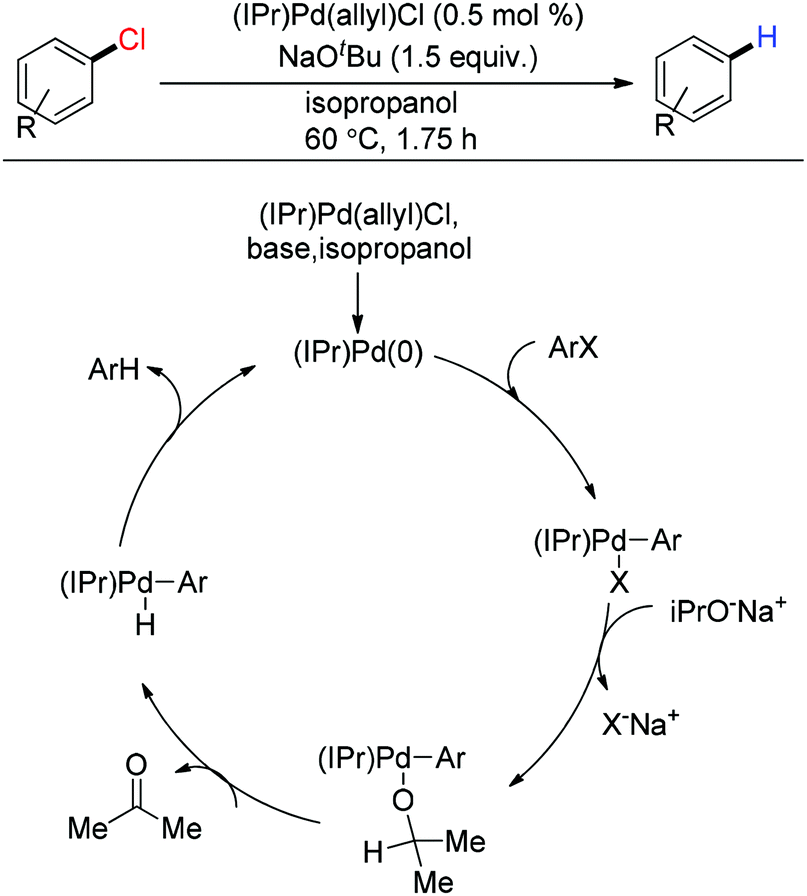
Palladium-catalysed Heck-type alkenylation reactions in the synthesis of quinolines. Mechanistic insights and recent applications - Catalysis Science & Technology (RSC Publishing)
![PDF] Palladium-triggered deprotection chemistry for protein activation in living cells. | Semantic Scholar PDF] Palladium-triggered deprotection chemistry for protein activation in living cells. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/3ae70a978b403098bfa58487a5f2c84d3e816087/3-Figure2-1.png)